AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Cobalt chrome melting point11/8/2023   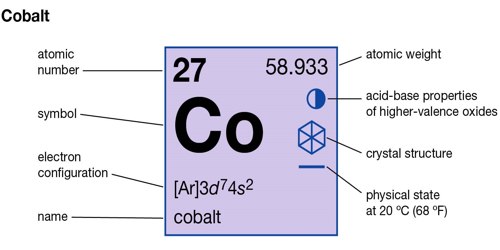
Those properties include a high strength-to-weight ratio, high tensile and yield points, high hardness, and great toughness. This oxide layer protects the alloy from environmental effects, helping it maintain its mechanical properties. Just like stainless steel, Cobalt Chrome develops its high corrosion resistance by forming a dense and passive oxide layer on its surface (i.e., Chromium oxide). The most commonly used alloys are cast/wrought Co28Cr6Mo and wrought Co35Ni20Cr10Mo. Cobalt Chrome alloys are generally classified into two categories: What is Cobalt Chrome (Cobalt Chromium alloy)?Ĭobalt Chrome, also known as Cobalt-Chromium or CoCr, is an alloy made mainly of Cobalt ( 53 – 67 %) and Chromium (25 – 32 %) with small added amounts of Molybdenum, Nickel, Tungsten, Silicon, or Aluminum for application-specific strengthening. In this article, we will look into the details of Cobalt Chrome, how it fares against other alloys, how it can be used in 3D printing, and where it is commonly used. Localized manufacturing, rapid prototyping, complex geometries, minimized material waste, and on-demand spare part production have all become possible for Cobalt Chrome with the advent of 3D printing. While maintaining the integrity and properties of Cobalt Chrome, additive manufacturing (AM) has facilitated and boosted its production and use. Metal 3D printing provided a stronger case for Cobalt Chrome. With high toughness, hardness, and strength-to-weight ratio, cobalt-chromium alloys became some of the most common materials in specific applications such as jet engines, gas turbines, and knee implants. It then became highly utilized in the medical industry mainly due to its biocompatibility. The specific type of cobalt alloy used to create cobalt rings is call cobalt chrome (CoCr).Cobalt Chrome is an alloy that was developed initially for the aerospace industry thanks to its excellent corrosion resistance and mechanical properties. The melting point of cobalt metal is 1,493☌ (2,719☏), and the boiling point is about 3,100☌ (5,600☏). It is also one of the only three naturally magnetic metal found on Earth, among iron and nickel. One of the variations of cobalt, called Cobalt-60 isotope, is used widely in treating cancer and killing cancer cells.Ĭobalt is very hard, very ductile and moderately malleable. Cobalt is also used in making of lithium batteries and chemical catalysts. 
The use of cobalt in coloring can be traced back to ancient Egypt and China civilizations. Other applications of cobalt include pigmenting glass and ceramic with green and blue color and manufacturing paint. High speed steel drill bits are also made from cobalt alloys due to their superior heat-resistance. Another reason that cobalt alloys are suitable for prosthetics part is being hypoallergenic as well. The corrosion and wear-resistant characteristics make cobalt based superalloys widely used in medical industries for replacing body parts such as knee and joint bones. The strength and stability of cobalt alloys make them the perfect material for building turbine blades and jet engines for aircrafts. Superalloys made from cobalt consume most of the cobalt mined worldwide. It can also be found in meteoric iron and vitamin B-12. Mostly yielded from Democratic Republic of the Congo, cobalt ore is mainly a by-product of copper and nickel mining. The word cobalt is taken from German word Kobold, which means “goblin”, a name used by miner to refer to cobalt ore. Cobalt, with chemical symbol Co and atomic number 27, is a lustrous grey metal and not found in its pure form in nature. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
